Sorry, that story no longer exists on our website
The article or video you are looking for has been moved to our archive. Perhaps another story on this page might interest you?
Scroll down for links to our latest news, crisis explainers and videos from our projects.
Latest news and stories from MSF UK

Israel’s manufactured malnutrition crisis in Gaza had devastating impacts on pregnant women and their newborns
Learn more

Although ceasefires have brought some relief and allowed MSF teams to scale up, the situation remains extremely fragile
Learn more

MSF calls on states including the UK to respect their commitment to protect medical care
Learn more
Never miss another story
Sign up to Frontline, our monthly e-newsletter, to stay up to date with MSF's life-saving work in over 65 countries
MSF crisis responses explained
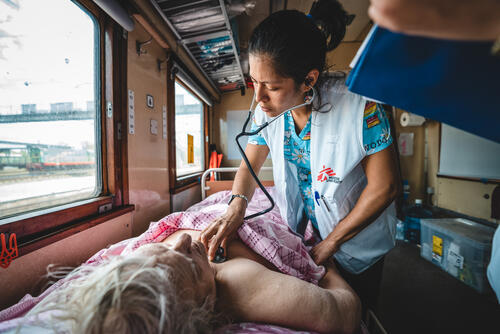
From the first days of the crisis, MSF has worked to deliver medical aid to people in Ukraine
Learn more

Yemen is in the middle of a complex humanitarian crisis driven by a brutal civil war
Learn more









